Title 1: Clinical Practice Guidelines for Inpatient Glycemic Control in Indian Healthcare Settings
Title 2: Guidelines for the Management of In-Hospital Hyperglycemia in India
Title 3: India-Specific Guidelines for Safe and Effective Management of Hyperglycemia in Hospitalized Patients
1 Process and methodology
2 Need for the recommendation
In-hospital hyperglycemia is common and associated with poor outcomes, prolonged hospital stays, higher readmission rates, increased need for transitional care, and elevated mortality [1, 2, 3]. Preoperative hyperglycemia, especially within 6 hours of surgery, predicts adverse outcomes [4]. Its prevalence ranges from 38–40% in hospitalized patients, 22–46% in non-critically ill [1, 5] , and up to 70–80% in critically ill or cardiac surgery patients [6, 7, 8, 1, 9]. Though often transient, up to 60% may develop diabetes within 6 months post-discharge [10, 11]. While managing hyperglycemia is vital, intensive insulin therapy can increase hypoglycemia and mortality risks [5, 12, 13, 14, 15, 16, 17, 18, 19, 20] [21, 22, 23]. Therefore, individualized glycemic targets are crucial. International guidelines offer adaptable strategies for patient-specific care, emphasizing safe drug choices, appropriate blood glucose monitoring, dosage adjustments, and nutritional considerations [24, 25, 26, 27]. These recommendations must be regularly reviewed and tailored to local disease patterns and healthcare settings to ensure optimal inpatient diabetes care.
3 Introduction and classification of in-hospital hyperglycaemia
India faces a rapidly escalating diabetes burden, with 74.9 million adults affected in 2021—projected to reach 124.9 million by 2045 [28]. Nationally representative studies, including the ICMR-INDIAB study [29], the National NCD Monitoring Survey (NNMS) [30], and NFHS-5 [31], show prevalence ranging from 7.7% to 16.1%, with significant geographic and socioeconomic disparities [32].
3.1 Definition of hyperglycaemia in a hospitalised patient
Hyperglycaemia in the hospital has been defined as any blood glucose level that is >140 mg/dl .
3.2 The burden of hyperglycaemia in the hospital setting
Global and national studies report in-hospital hyperglycemia prevalence ranging from 17.5% to 80%, depending on patient population, setting (ICU vs. non-ICU), and hyperglycemia criteria used [1, 9, 6, 8, 33, 34, 35]. In India, in-hospital hyperglycemia is a common complication affecting both diabetic and non-diabetic patients, with prevalence estimates ranging from 20% to 56% [29, 30, 31]. Among ICU patients, stress-induced hyperglycemia occurs in approximately 20% [30]. It is associated with increased mortality (OR: 2.5; 95% CI: 1.347–4.936; P=0.0063) [29] and worsens clinical outcomes due to impaired immunity and oxidative stress. Hospitalized older adults are particularly vulnerable. Effective, individualized glycemic control is crucial to improve outcomes and reduce healthcare burden.
This update provides context-specific guidance for inpatient diabetes care in India, addressing high rates of hyperglycemia and variability in clinical practice. It outlines monitoring protocols, glycemic targets, pharmacologic management, and care transitions, with special focus on older adults and critically ill patients to improve consistency and outcomes in Indian hospitals.
3.3 Classification of hyperglycaemia during hospitalisation
| Defining criteria | |
| Previously diagnosed diabetes | Existing hyperglycaemia prior to hospitalisation |
| Previously undiagnosed diabetes | At admission: Fasting plasma glucose (FPG) >126 mg/dl or Random blood glucose (RBG) >200 mg/dl or HbA1c >6.5% |
| Stress/Illness/infection induced hyperglycaemia | At admission: HbA1c <5.7% During hospitalization: FPG >126 mg/dl RBG>200 mg/dl Post discharge: Normoglycaemia |
4 Recognition of inpatient hyperglycaemia [5]
Accurately differentiating newly diagnosed diabetes from stress-induced hyperglycemia is crucial for appropriate management. Assessing glycemic control prior to admission helps establish whether elevated blood glucose reflects chronic diabetes or a transient stress response. This distinction guides clinical decision-making during hospitalization and enables the development of an optimal, individualized treatment regimen at discharge, ensuring effective long-term glycemic control and reducing the risk of complications. The details of monitoring of glycaemic status are covered in the respective sections of patient population.
4.1 Initial Laboratory Testing:
Plasma Glucose (PG) testing should be performed for all patients upon admission.
| Monitoring in Non-Diabetes Patients: |
|
|
| Monitoring Based on Risk Factors: |
|
|
|
|
|
| Monitoring in diabetes patients |
| HbA1c to be tested at admission [5]: Every hyperglycaemic hospitalised patient should have an HbA1c test done at admission if it has not been done in the last 2–3 months. |
4.1.1 Limitations of Bedside Glucose Monitoring
Be aware of variability: A CDC study showed up to 32% deviation from central lab values and a 6–11% coefficient of variation using commonly available glucose meters operated by a trained technologist.
Special Considerations in ICU Settings -
-
Glucose meter accuracy may be compromised by:
-
Hypotension or poor perfusion (e.g., in trauma or shock)
-
Hematocrit extremes
-
High oxygen therapy
-
Interfering medications: aspirin, paracetamol, vitamin C, dopamine, mannitol
Confirmatory Testing
If CBG results are inconsistent with the patient’s clinical status, confirm with laboratory plasma glucose testing.
5 Clinical practice recommendations for management of in-hospital hyperglycaemia can be further classified into care of the diabetic patient in the following three categories:
5.1 Targets: Guideline recommendations
The threshold for in-patient hyperglycaemia is defined as blood glucose level more than 140 mg/dl at the time of admission. Different guidelines have different target criteria for patients presenting with hyperglycaemia at admission (Table 2).
| 1. Critically ill (ICU) |
| 2. Non-critically ill (non-ICU) |
| 3. Special circumstances |
| Peri-operative conditions |
| Patients on enteral/parenteral nutrition |
| Patients on glucocorticoid therapy |
| Chronic kidney disease (CKD) patients |
| Transplant patients |
| Peri-partum situations |
| Guidelines | Patient population | Recommendations |
| Critically ill | ||
| ADA/ AACE [23] | Stress hyperglycaemia | 140-180 mg/dl |
| Hospital related hyperglycaemia | 140-180 mg/dl | |
| Select ICU patients cardiac surgery patients and patients admitted in centers with expert experience and nursing support) | 110-140 mg/dl | |
| Society of Critical Care Medicine [22] | ICU patients | <150 mg/dl |
| Non-critically ill patients | ||
| Endocrine Society guidelines [5] | Pre meal glucose Random blood glucose target |
<140 mg/dl <180 mg/dl |
| ADA/ AACE [23] | Pre meal glucose Random blood glucose target |
<140 mg/dl <180 mg/dl |
| ADA [33] | for most general medicine and surgery patients | 7·8 and 10·0 mmol/L |
| JBDS Inpatient Care group in the UK [34] | Random blood glucose target | 108–180 mg/dL |
| India recommendations [35] | Patients with new-onset hyperglycemia | 140-180 mg/dl |
| In cases with prediction of no risk of hypoglycemia | 110-180 mg/d | |
| Special considerations | ||
| Terminally ill patients, those with severe comorbidities, or in inpatient care settings where frequent glucose monitoring or close nursing supervision is not feasible. [33] |
Random blood sugar levels | <200 mg/dl |
Abbreviations: Association (ADA) and American Association of Clinical Endocrinologists (AACE)
In critical care setting, recommendation for a lower target is adopted while keeping hypoglycaemia under scrutiny. Although observational studies have observed mortality benefits with tighter glycaemic control, major associations recommend against a lower target blood glucose range for select ICU patients with increasing risk of hypoglycaemia and mortality. the Society of Critical Care Medicine recommends starting therapy for ICU patients with a blood glucose concentration of 8·3 mmol/L (150 mg/dL) or higher and maintaining glucose concentrations of less than 10·0 mmol/L (180 mg/dL) with strategies that minimise the risk of hypoglycaemia [22]. Therefore, a recent recommendation suggests that in an in-hospital setting, any patients critically ill have to be regularly monitored with a frequency of 4 times a day to attain a target blood glucose > 126 mg/dl. In terminally ill patients, a higher target of blood glucose <200 mg/dl may be desired and acceptable [36].
| Critically ill patients* | Non-critically ill patients | |
| General | <150 mg/dl | RBS 110-180 mg/dl |
| Diabetes | 140-180 mg/dl | |
| Diabetes with complications or comorbidities | ||
| Non-diabetes | ||
| Diabetes with complications or comorbidities | ||
| Stress hyperglycemia | 140-180 mg/dl |
*-terminally ill patients- target RBS <200 mg/dl
From previous publication
| Level of care | Glycemic target |
| ICU | 140 – 180 mg/dl |
| Non-ICU | Fasting PG 80 – 120 mg/dl |
| Pre-meal BG < 140 mg/dl | |
| Post-meal BG < 180mg/dl | |
| Special circumstances | Refer to the relevant section |
Glucose level of <110 or >180 mg/dl is not recommended
To achieve optimal glucose control in critically ill patients, it is essential to minimize the risks of both hyperglycemia and hypoglycemia through regular blood glucose (BG) monitoring. For patients receiving intravenous (IV) insulin, initial BG levels should be checked hourly until stabilization, after which monitoring can shift to every two hours. For those on oral feeds, BG should be measured at least four times daily—before meals and at bedtime. Changes in medication, such as the introduction of corticosteroids, may necessitate adjustments in monitoring frequency [32]. Additionally, older patients or those with comorbidities or a history of hypoglycemia may require more frequent and intensive monitoring to ensure safe glucose management [97].
Capillary glucose monitoring has long been the standard practice in intensive care units (ICUs) for managing insulin therapy. Recent evidence indicates that continuous glucose monitoring (CGM) is not only safe but also efficient, offering insights into glucose trends and fluctuations. However, most studies on CGM in critically ill patients have primarily focused on its accuracy rather than its clinical outcomes [98]. The accuracy of CGM can be influenced by factors such as tissue deposits around the sensor, which may affect readings [99]. Outcomes assessed in CGM research include the frequency and severity of hypoglycemic episodes and glycemic variability. The Normoglycemia in Intensive Care Evaluation-Survival Using Glucose Algorithm Regulation (NICE-SUGAR) trial highlighted that intensive insulin therapy (IIT) was associated with increased rates of hypoglycemia and higher mortality rates, underscoring the necessity for continuous glucose monitoring in critical care settings [14]. Similar findings have emerged from smaller studies involving specific patient populations. Despite these insights, there remains a need for extensive studies involving larger patient cohorts to fully evaluate the effectiveness of CGM in ICU environments. This gap in research is crucial for determining the broader applicability of CGM systems in managing glucose levels among critically ill patients [100].
Preferred In-hospital Monitoring Method
-
Bedside CBG is the preferred method for inpatient glucose monitoring.
Monitoring Frequency Based on Patient Status
-
Patients who can eat: Test before meals (not earlier than 1 hour before) and at bedtime.
-
Patients on continuous enteral nutrition (EN) or nil per os (NPO): Monitor every 4–6 hours.
-
Patients requiring more frequent monitoring:
-
On continuous insulin infusion (CII)
-
Receiving glucocorticoids
-
Undergoing abrupt discontinuation of EN or parenteral nutrition (PN)
-
Experiencing frequent hypoglycemia
For limitations and special considerations with monitoring in ICU, refer to section 4.1.1
Recommendations
| Among patients with or without diabetes, an increase in hypoglycemia increases adverse outcomes, length of ICU stay, and mortality. |
| It is important to adopt strategies to minimize hypoglycemia in critical care settings. |
| The minimum BG monitoring frequency should be at least four times a day. |
| More frequent and intense monitoring in case of: Clinical status, such as comorbidity chronic kidney disease (CKD), chronic liver disease (CLD), and elderly patients. Previous episode of hypoglycemia |
| Feedback loop: Nurse lead/computerized/hybrid system: none superior. |
6 Managing diabetes in critically ill patients in the ICU
Glycemic control in critically ill patients remains complex, with a general target of 140–180 mg/dL applied to both diabetic and non-diabetic individuals without persistent hyperglycemia. Exceptions exist for certain subgroups: stricter targets (110–140 mg/dL for neurological patients and <150 mg/dL for surgical patients) may be appropriate if hypoglycemia is avoided. Conversely, in patients with chronic hyperglycemia (HbA1c >7%), a more liberal approach may help minimize hypoglycemia and glycemic variability, although further research is needed to support firm recommendations [37].
Perioperative and Critical Care Glucose Management Guidelines [36, 36]
Initiation of Insulin Therapy:
Initiate insulin therapy when blood glucose (BG) >180 mg/dL (>10.0 mmol/L) in hospitalized patients (ADA/AACE, Critical Care Society) [38, 23].
Target Blood Glucose Ranges:
General ICU/Surgical/Medical Patients:
| ADA/AACE: Target BG 140–180 mg/dL (7.8–10.0 mmol/L) for most patients. |
| ACP: Target BG 140–200 mg/dL (7.8–11.0 mmol/L); avoid intensive insulin therapy regardless of diabetes status [16]. |
| Critical Care Society: Maintain BG <180 mg/dL (10.0 mmol/L), while avoiding hypoglycemia. |
Selected Patients (e.g., stable, without hypoglycemia risk):
| ADA/AACE: More stringent targets of 110–140 mg/dL (6.1–7.8 mmol/L) or even 100–180 mg/dL (5.6–10.0 mmol/L) may be appropriate. |
Surgical Patients (Society of Thoracic Surgeons) [38]:
| Recommend continuous insulin infusion over subcutaneous or bolus IV insulin. |
| Maintain intraoperative BG <180 mg/dL (<10.0 mmol/L). |
| Aim for BG ≤110 mg/dL (≤6.1 mmol/L) in fasting and pre-meal states. |
Although several algorithms exist for managing hyperglycaemia in critically-ill patients, overall protocol involves three crucial elements
Dynamic Rate Adjustment:
Insulin infusion rates should be adjusted based on the current and previous blood glucose (BG) values, as well as the current insulin infusion rate.
Trend-Based Decision Making:
Protocols should account for the rate and direction of BG change—whether increasing, decreasing, or stable—between readings to guide infusion rate modifications.
Frequent Glucose Monitoring:
Blood glucose should be monitored hourly until stable glycemia is achieved, then at least every 2 to 3 hours thereafter to maintain safe and effective control.
6.1 Preparation of insulin infusion (Table 2)
6.1.1 IV insulin infusion protocol
Starting an IV insulin infusion and give a priming bolus of regular insulin 0.1 U/kg body wt. if the initial BG >300 mg/dl.
Infusion initiation (U/h): Initial rate of infusion = current BG divided by 100 [11, 17]
Examples
| If >600 mg/dl | 500-600 | 400-500 | 300-400 | 200-300 |
| 6U/h | 5-6U/h | 4-5U/h | 3-4U/h | 2-3U/h |
In adjusting the insulin infusion rate, refer to Flowchart.
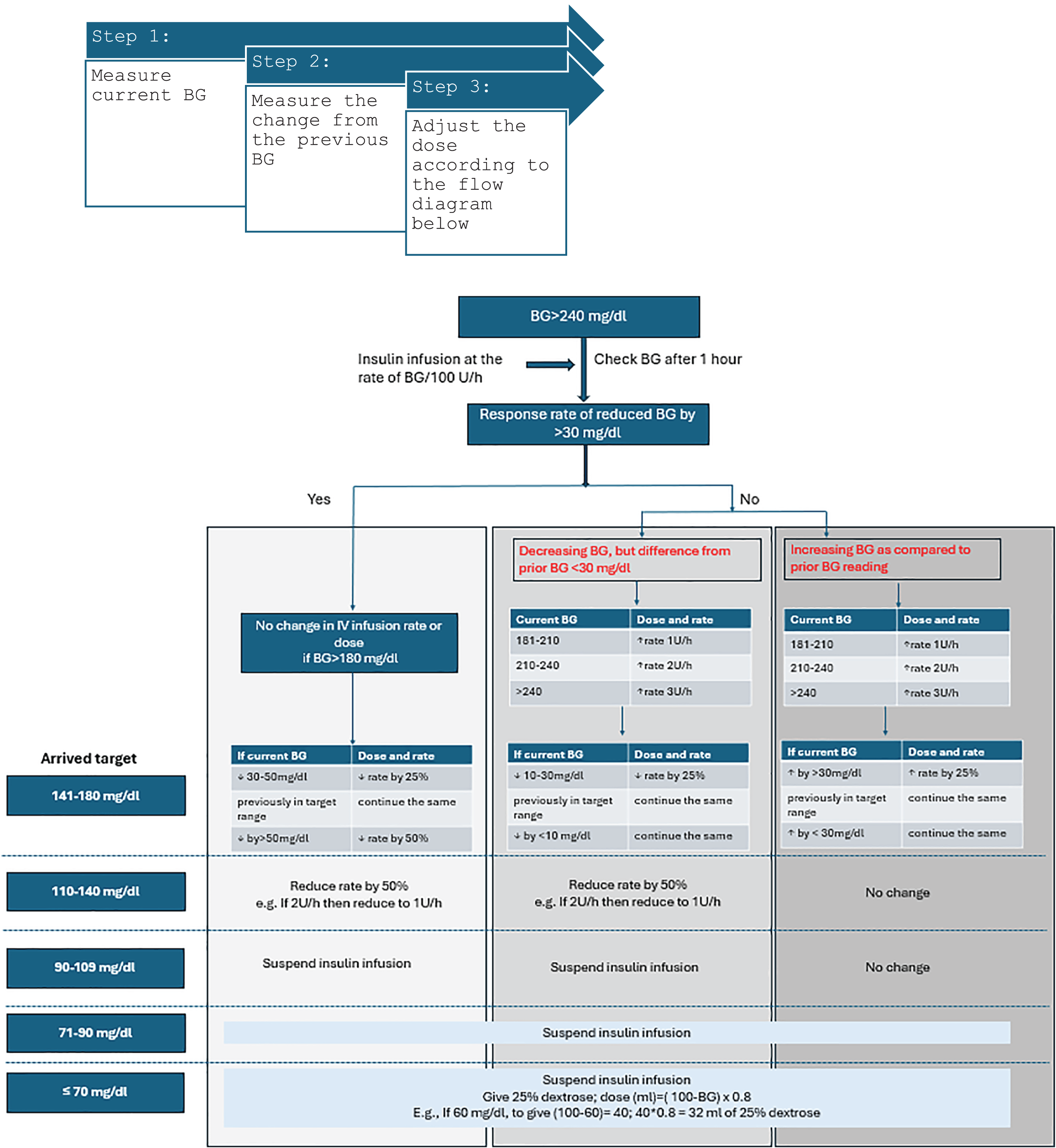 Figure 1
Figure 1
One hour after insulin initiation, three scenarios are possible
-
Favourable response rate of reduced BG by more than 30 mg/dl. In this scenario, the dose and rate could be continued until the BG is maintained between 140-180 mg/dl. However, if there is a steep drop in BG of 30-50 mg/dl or even more than 50 mg/dl, then the rate must be reduced by 25 and 50%, respectively. Also, if the BG is attained to 110-140 mg/dl, the rate is reduced by 50%. Insulin infusion must be suspended if BG is <110 and 25% dextrose must be considered if BG <70 mg/dl.
-
Decreasing BG but not the desired response of >30 mg/dl drop. In this scenario and with a BG >140 mg/dl , the rate has to be increased in accordance with the current BG status (flow chart).
7 Transition from insulin infusion to SC insulin and/or oral anti-diabetic drugs
Transitioning from IV to SC insulin is a crucial step in the management of hyperglycemia in hospitalized patients who no longer require intensive insulin therapy. This transition reduces mortality risk and lowers cost . Transitioning from IV to SC insulin requires a structured approach, including careful calculation of insulin needs, monitoring of blood glucose levels, and adjustments based on individual patient responses. This requires careful consideration of preventing rebound hyperglycaemia. There could be an increase in the hyperglycaemic events following transition [39].
The protocols involve calculating the total daily SC insulin requirement based on the IV insulin rate during the final 12 hours, dividing it into 50% basal and 50% prandial doses [40]. Among critically ill individuals, safe transition can be performed by transferring to 50-59% of their 24-hour IV insulin requirements [41].
Subcutaneous (SC) insulin administration in the non-ICU hospitalised patient should choose from the following options:
-
Basal insulin
-
Nutritional (bolus) insulin
-
Correctional insulin
-
NPH and regular insulin
-
Long-acting and rapid-acting insulin analogues
-
Pre-mixed insulin analogues
The use of incretin-based therapy with basal insulin with supplemental rapid-acting insulin needs more research. For most patients in the hospital, basal bolus is the preferred regime. However, if patients have been well controlled on pre-mixed insulin, they can be continued on the same regime. These patients require frequent monitoring [35].
7.1 Estimation of dose in SC insulin injections
Patients, who were well controlled on insulin prior to admission, can usually be maintained on their home insulin regimen with specific adjustments made for differences in meals and activity levels, the effects of illness and the effects of other concomitant medications.
In patients who were on an insulin infusion and now transferred to non-critical care settings, the TDD of SC insulin can be calculated from the insulin requirement in previous 6-8 h of stable control of BG and multiplied by 4 to get the TDD in 24 h or the actual total insulin requirement in the last 24 h [42, 43]. Alternatively, weight-based approach shall be considered. For insulin-naive patients or when no IV insulin therapy has been given, the TDD of insulin can be calculated as shown in Tables below [35].
7.2 . Timing of Transition
-
Clinical Indication: Transition once the patient's clinical status stabilizes and blood glucose levels have been within target range for at least 12–24 hours.
-
Evidence: Low-dose basal insulin analog (0.15–0.3 units/kg) co-administered with IV insulin can prevent rebound hyperglycemia and reduce hospital stay [44, 45, 46].
7.3 . Choice of Insulin Regimen
-
Recommended Regimen: Basal-bolus regimen using long-acting (e.g., glargine or detemir) and rapid-acting (e.g., aspart or lispro) insulin analogs.
7.4 . Insulin Dosing Strategy
Calculation Method: Base the total daily dose (TDD) on prior 6–8 hours of IV insulin or weight-based calculation [47].
Dosage Adjustment: Administer 75–80% of estimated TDD as SC insulin.
Example: If IV rate = 20 units/hr, TDD = 480 units; SC insulin = 360–400 units/day, split into basal and bolus.
Weight-based basal insulin (0.2–0.25 units/kg) during IVII and initiating a basal-bolus regimen post-infusion improves glycemic control at 24 hours without increasing hypoglycemia risk, compared to standard care.
This strategy supports initiating basal insulin coverage during IVII, followed by a structured basal-bolus transition to maintain glycemic stability after discontinuing IV insulin.
Sample Calculation:
For a patient weighing 70 kg:
Basal Insulin Dose During IVII:
0.2–0.25 units/kg × 70 kg = 14–17.5 units of basal insulin (e.g., glargine or detemir)
Transition to Basal-Bolus After IVII:
Total daily dose (TDD): Start with 0.4–0.5 units/kg × 70 kg = 28–35 units/day
Basal (50% of TDD): 14–17.5 units once daily
Bolus (divided before meals): 14–17.5 units split across 3 meals (~5–6 units per meal)
Adjustments should be individualized based on nutritional intake, renal function, and glucose trends.
7.5 Transitioning from IV to SC Insulin: Practical Considerations
The use of insulin pumps may be considered in patients requiring precise glucose control, especially those previously managed with continuous subcutaneous insulin infusion at home. These devices allow for continuous basal insulin delivery and flexible bolus dosing, supporting individualized glycemic targets.
Following the transition from intravenous (IV) to subcutaneous (SC) insulin, close monitoring is essential to ensure optimal glycemic control. Blood glucose should be monitored every 4–6 hours during the first 24 hours post-transition. In high-risk patients, continuous glucose monitoring (CGM) may be beneficial. The American Diabetes Association (ADA) 2024 guidelines recommend tailoring the monitoring frequency based on individual patient response and risk profile.
Several challenges can arise during this transition phase. Common pitfalls include delayed or inappropriate insulin dosing, fluctuating blood glucose levels, renal dysfunction, infections, and concurrent corticosteroid use. These factors necessitate timely and dynamic adjustments of insulin doses according to clinical parameters and evolving meal patterns.
Patient education plays a pivotal role in successful transition and long-term diabetes management. Educational efforts should focus on insulin self-administration, recognizing and managing symptoms of hypo- and hyperglycemia, and understanding how to adjust doses as needed. Ideally, education should begin early during the hospital stay, particularly for patients who will be discharged on outpatient SC insulin therapy, to ensure they are well-prepared and confident in managing their condition.
7.6 Transition from subcutaneous insulin to oral drugs
Transitioning from subcutaneous insulin to oral antidiabetic medications in hospitalized patients with hyperglycemia is a critical process that requires careful consideration of the patient's glycemic control and overall clinical status. Clinical guidelines suggest that transitioning to oral agents should occur only after confirming that the patient can maintain glycemic control without the need for intensive insulin therapy. For instance, in patients who require only very low dose of insulin to maintain good glycemic control, continuous monitoring of blood glucose levels during this transition is essential to avoid episodes of hyperglycemia or hypoglycemia. Additionally, patient education regarding the new medication regimen and its administration is vital to ensure adherence post-discharge.
7.6.1 Incorporating Oral Agents with Basal Insulin in Hospitalized Patients
Refer to section 8.2 for the appropriate use of oral anti-glycemic agents in an in-hspital setting
| Summary of Key Recommendations |
| Use basal-bolus SC regimens based on IV insulin data. |
| Reduce SC insulin dose by 20–30% of IV TDD. |
| Monitor glucose every 4–6 hours post-transition. |
| Consider CGM in high-risk patients. |
| In selected patients, augment basal insulin with oral agents (e.g., SGLT2i, GLP-1RA, DPP-4i). |
| Initiate patient education early. |
8 Glycaemic control in the non-critically ill patient
-
Insulin is the preferred treatment for hyperglycaemia in hospitalized patients with diabetes
-
Patients with T1DM must be maintained on insulin therapy at all times to prevent DKA
-
Glycemic Targets are pre-meal BG <140 mg/dl and post meal BG <180 mg/dl
Inpatient Glycemic Targets in Non-Critical Settings- existing guideline recommendations
Inpatient Glycemic Targets: Comparison of Major Guidelines
| Guideline | Initiation Threshold | General BG Target Range | Stricter Target (if safe) | Lenient Target (special cases) | Additional Notes |
| ADA/AACE ( [23, 38] Refs 20, 108) |
Initiate insulin if BG ≥180 mg/dL (10.0 mmol/L) | 140–180 mg/dL (7.8–10.0 mmol/L) | 110–140 mg/dL (6.1–7.8 mmol/L) for selected post-op or cardiac ICU patients | >250 mg/dL (13.9 mmol/L) acceptable in end-of-life or severe comorbidities | Emphasizes individualized goals |
| Endocrine Society [5, 25] (Refs 19, 109) |
— | 100–180 mg/dL (5.6–10.0 mmol/L) in insulin-treated patients | Pre-meal <140 mg/dL (<7.8 mmol/L) | 180–200 mg/dL (10.0–11.0 mmol/L) in terminal illness or limited life expectancy | Adjust if BG <100 mg/dL (<5.6 mmol/L) |
| Joint British Diabetes Society (Ref 111) |
— | 108–180 mg/dL (6.0–10.0 mmol/L) for most inpatients | — | 72–216 mg/dL (4.0–12.0 mmol/L) as acceptable based on context | Allows broader flexibility |
Evidence-Based Recommendations for Subcutaneous Insulin Use in Hospitalized Patients
Scheduled subcutaneous insulin therapy using basal insulin (e.g., glargine, detemir, degludec once daily or NPH twice daily) combined with short- or rapid-acting insulin (e.g., regular, lispro, aspart, glulisine) before meals is effective and safe for most hospitalized patients with hyperglycemia and diabetes (20; 108; 194).
8.1 Insulin initiation
Subcutaneous (SC) insulin administration in the non-ICU hospitalised patient should include three components to be effective:(195)
-
Basal insulin- for fasting glucose control
-
Nutritional (bolus) insulin- for prandial glucose disposal
-
Correctional insulin- supplemental for correction of hyperglycemia
Alternative to Basal-Bolus – "Basal Plus" Regimen:
In patients with reduced caloric intake, insulin-naive individuals, or those previously on low-dose insulin (≤0.4 U/kg/day), the basal plus regimen (basal insulin once daily + corrective rapid-acting insulin for BG >140 mg/dL) offers comparable glycemic control and hypoglycemia risk as the full basal-bolus approach (108; 197). Other insulin regimens include:
-
Basal/ bolus therapy
-
NPH and regular insulin
-
Long-acting and rapid-acting insulin analogues
-
Pre-mixed insulin analogues
-
Use of incretin-based therapy with basal insulin with supplemental rapid-acting insulin as required
-
Glucagon-like peptide-1 receptor agonist (GLP-1 RA) in hospitalised patients- as they can reduce the total daily dose of insulin and minimise the risk of hypoglycaemia. They can be used if there are no contraindications to incretins.
8.1.1 Estimating SC insulin dose
Patients, who were well controlled on insulin prior to admission, can usually be maintained on their home insulin regimen with specific adjustments made for differences in meals and activity levels, the effects of illness and the effects of other concomitant medications.
In patients who were on an insulin infusion and now transferred to non-critical care settings, the TDD of insulin can be calculated from the insulin requirement in previous 6 h of stable control of BG and multiplied by 4 to get the TDD in 24 h or the actual total insulin requirement in the last 24 h.
Recommended Starting Total Daily Insulin Dose
For most individuals with diabetes, initiate insulin at 0.3 to 0.5 units/kg/day (139; 147; 198; 199).
Dosing Considerations to Reduce Hypoglycemia Risk:
Avoid starting doses >0.6–0.8 units/kg/day, as they are associated with a 3-fold increased risk of hypoglycemia compared to starting doses <0.2 U/kg/day.
In elderly patients or those with renal impairment, use a lower starting dose (≤0.3 units/kg/day) to minimize hypoglycemia risk (200).
8.1.2 Insulin dose modification in hospital
In patients who are NPO or unable to eat, bolus insulin must be withheld until nutrition is resumed. However, doses of correction insulin can be continued to treat BG above the desired range.
Adjustments of scheduled basal and bolus insulin can be based on total doses of correctional insulin administered in the previous 24 h.
| TDD calculation | Dose 80% of Calculated TDD | |
| For insulin naïve | Total dose as per body weight | Use 80% of calculated TDD for starting SC insulin |
| Ideal body weight | 0.4 – 0.5 U / kg | Given when |
| Obese | 0.5 – 0.6 U / kg | Basal insulin is 50% |
| Lean or renal compromised | 0.3 – 0.4 U / kg | Basal insulin is 50% |
| From IV to SC transition | ||
| Calculate the insulin requirement in the previous 6 hours of stable control of BG and multiply by 4 to get TDD in 24 hours or the actual total insulin requirement in the last 24 hours | ||
| Basal dose | Correctional bolus (CB) | |
| Glargine | Single dose AM/PM | BG – 100 = CB |
| Detemir | Given in 12-hours split dose | CF |
| NPH | Given in 12-hours split dose | CF = 1500 / TDD (regular insulin) |
| For basal dose correction | ||
| PM basal: check FPG every day, increase 2 U of basal till FPG of 80 – 120 mg/dl is achieved | ||
| AM basal: check pre-dinner BG every day, increase 2 U of basal till BG < 140mg/dl (for BD dose of NPH/deteminir | ||
| Bolus dose | ||
| Total bolus dose/3 With breakfast lunch and dinner | ||
| For Bolus dose regular, aspart, glulisine or lispro can be used | ||
SSI- Sliding scale insulin as a sole regimen for the management of hyperglycemia is ineffective and not recommended (109; 190-193)
8.2 Role of oral anti-diabetic drugs/non-insulin injectables in non-critically ill patients
Contraindications with use of OADs and other non-insulin therapies- Sepsis, NPO, IV contrast dye, pancreatic disorders, renal failure
Patients who are clinically stable and eating regular meals and have no contraindications to the use of these agents may continue their previously prescribed OAD therapy in the hospital
Use of Non-Insulin Glucose-Lowering Agents in Hospitalized Patients
While insulin remains the cornerstone for managing hyperglycemia in hospitalized patients, non-insulin therapies are often encountered due to pre-admission use or select clinical considerations. However, their utility in the inpatient setting is restricted by concerns regarding safety, variable pharmacodynamics, delayed onset, or contraindications in acute illness.
Metformin, the first-line agent in outpatients, poses risks in hospitalized patients with dehydration, renal dysfunction, or prior to contrast administration due to the potential for lactic acidosis. Thiazolidinediones (TZDs), though effective in the outpatient setting, are seldom appropriate acutely due to their slow onset and risk of exacerbating fluid overload and heart failure. Sulfonylureas (SUs), although still used in several countries, are associated with a high risk of hypoglycemia, particularly in elderly or renally impaired patients.
SGLT-2 inhibitors lack sufficient data for routine inpatient use but may be cautiously continued in select heart failure patients post-stabilization. SGLT2 inhibitors have limited evidence for routine inpatient use. Recent studies in acute illness, such as COPD exacerbations and COVID-19, failed to show outcome benefits (210–213). However, in people hospitalized for heart failure, the ADA recommends initiating or continuing SGLT2 inhibitors (e.g., empagliflozin) if there are no contraindications and once acute illness resolves. These agents should be withheld in type 1 diabetes, during prolonged fasting or surgical procedures, or in the presence of ketonemia/ketoacidosis. The FDA advises discontinuation at least three days before surgery (four for ertugliflozin), while UK guidelines recommend stopping one day prior (218–223).
Meanwhile, DPP-4 inhibitors have shown efficacy and safety in mild to moderate hyperglycemia in non-ICU patients, making them viable in combination with basal insulin. GLP-1 receptor agonists are limited by gastrointestinal side effects and insufficient evidence for inpatient use.
| Drug Class | Use in Hospital Setting | Common Risks / Limitations | Evidence / References |
| Sulfonylureas | Used in some inpatients, mostly continued from pre-hospital therapy | - Severe and prolonged hypoglycemia, especially in elderly and renal-impaired patients - Skipping meals increases hypoglycemia risk - Long-acting SUs may have challenging dose adjustments |
(140, 141, 203, 205–208) |
| Metformin | Generally avoided during hospitalization; restart post-stabilization | - Risk of lactic acidosis in dehydration, renal impairment, hypoperfusion, or chronic pulmonary disease - Must be discontinued before IV contrast or major surgery - Restart only if renal function is normal after 48h |
(195, 201) |
| Thiazolidinediones | Rarely used due to slow onset and risk of fluid retention | - Delayed onset (weeks) - Contraindicated in CHF, hepatic dysfunction, hemodynamic instability - May continue if already on therapy and stable |
(202–204) |
| DPP-4 Inhibitors | Effective for mild-to-moderate hyperglycemia in non-critical patients | - Safe as monotherapy or with basal insulin - Useful alternative to basal-bolus in selected patients - Contraindicated in history of pancreatitis, medullary thyroid carcinoma, or gastroparesis |
(48, 214–217) |
| GLP-1 Receptor Agonists | Not routinely used; under investigation | - GI side effects: nausea, vomiting - Risk of volume depletion - Contraindicated in pancreatic disorders, MTC, gastroparesis - No clear benefit in inpatient glucose control |
(Limited inpatient evidence) |
| SGLT-2 Inhibitors | Consider continuation in heart failure patients after stabilization | - Risk of genitourinary infections - Euglycemic DKA - Avoid in T1DM, fasting, or perioperative period - Insufficient benefit in acute COPD/COVID illness - Requires diuretic adjustment - Stop 3 days before surgery (4 for ertugliflozin) |
(210–213, 218–223) |
Limited care setting: Human insulin rather than analogues may be used
9 Management In Special Circumstances
9.1 Steroid-induced hyperglycemia
Steroid-induced hyperglycemia (SIHG) occurs when glucocorticoid therapy leads to elevated blood glucose levels, potentially causing new-onset hyperglycemia or worsening control in patients with existing diabetes. The condition arises due to the steroids' effects on insulin resistance and glucose metabolism, often exacerbated by factors such as the dose, duration of treatment, and patient predispositions like obesity or a family history of diabetes [48]. Studies indicate that approximately 32.3% of patients receiving glucocorticoids may experience hyperglycemia, with a significant portion requiring management strategies tailored to their specific needs [49]. Management of SIHG typically involves close monitoring of blood glucose levels and may necessitate adjustments in diabetes medications or initiation of insulin therapy, especially in hospitalized patients. For those on high-dose or prolonged steroid regimens, careful titration and monitoring are crucial to prevent complications associated with both hyperglycemia and hypoglycemia [48].
Recommendation
Management of patients with GC-associated hyperglycemia requires ongoing BG monitoring with adjustment of insulin dosing. All therapies require safeguards to avoid hypoglycemia when doses of GCs are tapered or abruptly discontinued. It is recommended to closely monitor blood glucose levels and adjust insulin doses accordingly to manage steroid-induced hyperglycemia effectively.
9.2 Diabetes-related complications
Post-surgery, it is crucial to closely monitor patients for the development of foot ulcers, kidney disease, and vision changes, as these complications can significantly impact recovery and overall health outcomes. Foot ulcers, particularly in diabetic patients or those with impaired circulation, can lead to infections and prolonged hospital stays if not identified early [50]. Additionally, kidney function should be regularly assessed, as surgical stress and medications can affect renal health, potentially leading to acute kidney injury [51]. Vision changes may also arise due to various factors, including medication side effects or underlying conditions exacerbated by surgery [52]. By implementing vigilant monitoring for these issues, healthcare providers can facilitate timely interventions, enhancing patient safety and promoting a smoother recovery process.
Recommendation
Monitor patients post-surgery for foot ulcers, kidney disease, and vision changes to ensure early detection and management of potential complications.
10 Special Considerations
10.1 Diabetic ketoacidosis (DKA)
Decompensated diabetes represents a significant challenge, impacting both economic factors and patient outcomes. Two critical conditions within this spectrum are diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS), both of which contribute greatly to morbidity and mortality in individuals with diabetes, even with established diagnostic guidelines and treatment protocols [53]. DKA is characterized by a biochemical triad that includes hyperglycemia, ketonemia, and a high anion gap metabolic acidosis, while HHS may exhibit varying degrees of clinical ketosis and alterations in consciousness, often without progression to coma. Both DKA and HHS are marked by hyperglycemia and varying levels of insulin deficiency. They differ mainly in terms of dehydration severity, the presence of ketosis, and metabolic acidosis levels. The leading triggers for these conditions are typically inadequate insulin therapy or infections [54]. Other factors contributing to their onset can include serious events such as myocardial infarction or pulmonary conditions, as well as certain medications like corticosteroids and diuretics [55]. Recently, SGLT-2 inhibitors, a newer class of diabetes medications, have also been associated with a heightened risk of DKA, particularly affecting patients with Type 1 and Type 2 diabetes. Furthermore, immune checkpoint inhibitors used in cancer treatment have been linked to new-onset diabetes, with a notable portion of those cases presenting as DKA [56, 57].
The pathophysiology of both DKA and HHS involves several key defects: reduced insulin effectiveness, elevated counter-regulatory hormones, and dehydration accompanied by electrolyte imbalances primarily caused by osmotic diuresis. Notably, DKA is associated with enhanced gluconeogenesis, lipolysis, and ketogenesis, while glycolysis is decreased [55].
Treatment goals for patients experiencing hyperglycemic crises include improving circulatory volume, gradually lowering serum glucose and osmolality, correcting electrolyte imbalances, and addressing any co-existing precipitating factors. Initial fluid therapy aims to expand intravascular volume, alongside the administration of insulin once serum potassium levels are deemed safe, specifically above 3.3 mmol/L [58, 55]. For DKA, an initial intravenous bolus of regular insulin is recommended, followed by a continuous infusion, whereas insulin administration in HHS may proceed at a slower rate due to the severe dehydration present [55].
In cases of DKA with significant hypokalemia, potassium replacement should accompany fluid therapy, delaying insulin treatment until potassium levels rise above the critical threshold to mitigate risks such as arrhythmias. For detailed management plans, consulting the latest DKA and HHS treatment guidelines is advised [58, 55].
Prior to undertaking any elective surgery, it is crucial to diligently address and manage diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS). This comprehensive approach entails a proactive regimen of fluid and electrolyte management, aimed at restoring equilibrium within the body [55]. Achieving a balanced acid-base status is also vital, which may necessitate the careful administration of bicarbonate alongside insulin to facilitate metabolic corrections. Throughout this meticulous process, vigilant monitoring of potential kidney overload is essential, as it plays a significant role in safeguarding overall health and ensuring optimal surgical outcomes [59]. In extremely rare instances, when DKA presents as life-threatening, intensive treatment must be sustained. Once the patient is stabilized, it is advisable to defer surgery to a later date. In exceptional cases, surgery may be performed concurrently with DKA management; however, potential complications such as blood coagulation issues and sepsis must be closely monitored [60]. Since it is beyond the scope to delve into the details of the management of this complex condition, we direct the practitioner to refer to a comprehensive DKA protocol for detailed instructions.
Recommendations
Before any elective surgical procedure, it is crucial to address DKA and HHS through aggressive management of fluids and electrolytes, as well as acid-base balance, which may include bicarbonate infusion and insulin administration.
Continuous monitoring for kidney overload is essential throughout this process.
10.2 Continuous renal replacement therapy (CRRT)
The effects of kidney dysfunction, which impacts glucose metabolism, as well as the uremic environment that arises from the accumulation of waste products in the body. The kidneys prevent hyperglycemia through the reabsorption of filtered insulin and increasing of urinary glucose excretion, while preventing episodes of hypoglycemia by the reabsorption and production of glucose via gluconeogenesis. Consequently, the risks of hypoglycemia increase in the advanced stages of DKD, with a direct correlation with the duration of diabetes, advanced age, polypharmacy and progression of renal failure [61, 62].
Dialysis-associated hyperglycemia (DAH), is associated with a distinct fluid and electrolyte pathophysiology.
This includes
-
Hypoglycaemia as a consequence of insulin treatment.
-
Elevated serum tonicity
-
Volume abnormalities ranging from pulmonary edema to osmotic diuresis led hypervolemia
-
Acid-base disorders, including ketoacidosis
-
Hyperkalaemia either at presentation or developed later during insulin infusion.
Insulin Therapy and dosage adjustments
The blood glucose levels of the patients on dialysis need to be monitored regularly. Insulin infusion remains the primary management strategy in DAH and corrects for most of the fluid and electrolyte imbalance [63].
Management of insulin in patients with varying stages of Diabetic Kidney Disease (DKD) requires careful dose adjustments. As renal function declines, it is usually necessary to progressively reduce insulin doses. For individuals without kidney issues, the recommended insulin doses range from 0.3 to 1 unit/kg/day for type 1 diabetes and 0.1 to 0.2 units/kg/day for type 2 diabetes, with both types typically comprising about 50% basal (long-acting) insulin and 50% prandial (fast-acting or ultra-fast acting) insulin [64].
For patients starting dialysis, significant reductions in insulin doses are crucial, especially on treatment days. All types of insulin generally require personalized dose adjustments when GFR falls below 60 mL/min, regardless of any pharmacokinetic changes. Patients on dialysis particularly need careful dose modifications, and monitoring fasting capillary blood glucose is recommended before dialysis, two hours into dialysis, at the end of the session, and two hours after completion.
In peritoneal dialysis settings, research indicates that a Glycosylated Hemoglobin A1c (GA) level greater than 20.0% is associated with decreased survival rates, primarily due to infections [65]. Achieving metabolic control is essential, and insulin therapy plays a critical role. Insulin can be administered subcutaneously or intraperitoneally, although the latter often requires higher doses due to losses in the dialysate and absorption onto device materials. A meta-analysis suggested that intraperitoneal insulin might need to be administered at double the amount compared to subcutaneous insulin to achieve better glycemic control [66]; however, this method carries a higher risk of bacterial contamination and hepatic subcapsular steatosis in diabetic patients, leading to increased insulin needs, obesity, and elevated triglyceride levels [67]. Further research is necessary to evaluate the long-term safety and effectiveness of intraperitoneal versus subcutaneous insulin in peritoneal dialysis, with subcutaneous administration generally being preferred.
Dialysis patients often have misleadingly low HbA1c due to factors like shortened red blood cell lifespan. Glycated albumin (GA), is suggested as a more reliable marker, correlating with mortality risk [68]. Generally, for an optimal glycemic control to minimize risks of hypo- and hyperglycemia, especially during periods of dialysis and after meals, maintain the target blood glucose range of 140-180 mg/dL. Regularly review and modify targets as appropriate based on patient circumstances and clinical guidelines.
Recommendations
Monitoring: Check BG every 6 hours.
Insulin therapy: Initiate or continue SSI or basal-bolus insulin therapy.
Dose adjustment: Adjust insulin doses based on BG trends and dialysis schedule.
Target BG range: 140-180 mg/dL
Oral anti-glycemic agents
The use of insulin therapy in hospitals is common in the USA and Canada [69, 26]; however, this practice varies internationally. While clinical guidelines generally recommend discontinuing oral antidiabetic drugs during hospitalization, in countries such as the UK, India, and Israel [70, 71], non-insulin agents like metformin are frequently utilized. Clinical trials have indicated that non-insulin drugs, whether used alone or in combination with basal insulin, can effectively achieve glycemic control for selected populations.
In the context of preoperative to perioperative care, sulfonylureas (SUs) should be avoided due to a significant risk of hypoglycemia. Additionally, SGLT2 inhibitors should not be given alongside insulin due to their potential to cause both hypoglycemia and urinary tract infections (UTIs). During the pre-operative phase, it's essential to withdraw oral antidiabetic drugs (OADs) that pose potential risk for hypoglycemia and ketoacidosis. Therefore, transitioning to insulin is crucial for managing blood sugar levels. This guideline is not important for minor surgical procedures, such as cataract or hernia surgeries, where the management of diabetes can still be essential yet manageable. If the patient remains stable during the perioperative period, insulin is recommended to be continued. After stabilization, a transition back to oral antiglycemic agents and combinations can be considered. Again, the key points are to stop SUs due to hypoglycemia risk, avoid using SGLT2 inhibitors with insulin, and manage the transition to insulin carefully.
10.3 Glycemic management guidelines for patients on steroids, vasopressors, unconscious, parenteral feeding, transplant, and dialysis
Recommendations
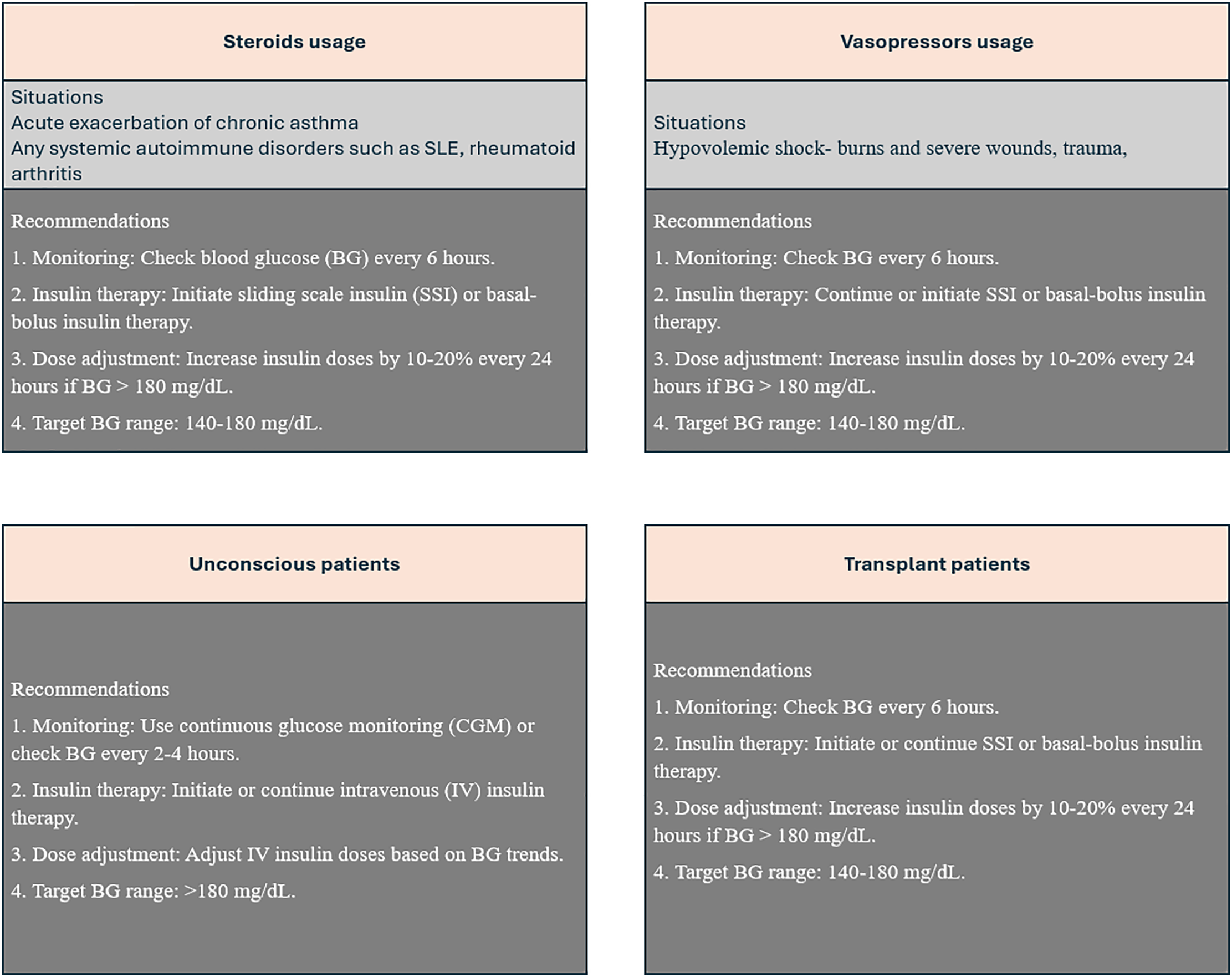
General Recommendations
Individualize therapy: Customize glycemic management strategies to address the unique needs and circumstances of each patient, considering their medical history, lifestyle, and personal preferences to achieve optimal outcomes.
Monitor electrolytes: Conduct regular and thorough assessments of electrolyte levels, with a particular focus on potassium and magnesium, as imbalances can significantly impact overall health and complicate glycemic control.
Adjust medications: Carefully evaluate and modify medications that may influence blood glucose levels, including beta-blockers and select antibiotics, to mitigate any adverse effects and enhance the effectiveness of diabetes management.
Hypoglycemia management: Develop a comprehensive and proactive plan for managing hypoglycemia, ensuring that patients are well-informed about the signs and symptoms, and are equipped with the necessary tools, such as glucagon and glucose, to swiftly address low blood sugar episodes.
11 Nutrition and Meal Planning in Hospitalized Patients with Hyperglycemia
Medical Nutrition Therapy (MNT) should be a core component of glycemic management in hospitalized patients with diabetes or hyperglycemia. [1] In the inpatient setting, the management of medical nutrition therapy (MNT) is typically guided by clinical judgment and individualized patient requirements, as robust evidence on optimal strategies remains limited. Determining the appropriate caloric intake and method of delivery should involve close collaboration between the healthcare team and a qualified nutrition specialist [72].
Nutritional plans must be individualized, considering age, reason for admission, BMI, comorbidities, and clinical status. [2]. Since hospitalization often induces a catabolic state, especially in critically ill patients and those undergoing surgery, adequate protein intake must be ensured. Additionally, patients on basal insulin may require periodic snacks—midmorning, midday, evening, and at bedtime—to prevent hypoglycemia.
Nutritional Needs in Hospitalized Patients with Diabetes
In the inpatient setting, most individuals with diabetes can meet their nutritional needs through three structured meals per day. Caloric requirements generally range from 25–35 kcal/kg/day, with critically ill patients requiring a more conservative intake of 15–25 kcal/kg/day (250; 251). A consistent carbohydrate meal-planning approach is recommended to support glycemic control and facilitate insulin dosing. Carbohydrate intake should be distributed evenly, ideally from low glycemic index sources such as whole grains, legumes, and vegetables (19).
| Nutritional Parameter | Target / Recommendation | References |
| Caloric requirement – General | 25–35 kcal/kg/day | (250; 251) [73, 74] |
| Caloric requirement – Critically ill | 15–25 kcal/kg/day | (250; 251) [73, 74] |
| Daily energy intake – Most adults | 1,500–2,000 kcal/day | (19) [5] |
| Carbohydrate per meal | 12–15 grams | (19) [5] |
| Mean frequency | Three structured meals/ day |
Nutritional Requirements
-
Caloric intake: ~25 kcal/kg/day
-
Carbohydrates: ~200 g/day
-
Protein: ~0.8–1.0 g/kg/day (higher in catabolic states)
-
Adjust macronutrients based on clinical context and route of nutrition
11.1 Oral Nutrition
-
Use consistent carbohydrate meal plans synchronized with insulin therapy
-
Provide three structured meals per day, with mid-meal snacks if required [2]
-
Emphasize low glycemic index foods to reduce postprandial glucose spikes
-
Avoid sugar-rich options (e.g., sweetened drinks, fruit juices); prefer whole fruits and healthy fats [4]
Enteral and Parenteral Nutrition in Hospitalized Patients with Diabetes
When oral intake is inadequate, assess the need for enteral (EN) or parenteral nutrition (PN). EN is preferred due to lower infection and thrombotic risk (254–256). Nutrition support helps prevent malnutrition and catabolism (252).
Standard formulas provide 1–2 kcal/mL and include whole protein, long-chain triglycerides, and 55–60% of calories from carbohydrates. Diabetes-specific formulas are lower in carbohydrates, higher in monounsaturated fats (up to 35%), and contain 10–15 g/L fiber and up to 30% fructose. These LCHM formulas reduce postprandial glucose by 18–29 mg/dL (1.0–1.6 mmol/L) compared to standard formulas (257, 258).
| Component | Standard Formula | Diabetes-Specific (LCHM) | References |
| Energy Density | 1–2 kcal/mL | 1–1.5 kcal/mL | (257) |
| Protein | Whole protein | Whole protein | (257) |
| Fat | LCTs (25–30% kcal) | MUFAs (up to 35% kcal) | (257, 258) |
| Carbohydrates | 55–60% of total kcal | Lower; partially replaced by MUFAs | (257, 258) |
| Fiber | Low | 10–15 g/L | (257) |
| Fructose | Minimal | Up to 30% | (257, 258) |
| Glycemic Impact | Higher postprandial glucose rise | ↓ 18–29 mg/dL (1.0–1.6 mmol/L) | (258) |
11.2 Enteral Nutrition (EN)
For continuous enteral feeds via Ryle’s tube or jejunostomy, insulin is administered using a basal-bolus regimen with multiple subcutaneous injections.
|
|
|
|
|
|
|
|
|
|
11.3 Parenteral Nutrition (PN)
-
Initially, a separate IV infusion of regular insulin may be needed to estimate the total daily dose (TDD)
-
This shall be stopped and 80% of the 24-hour dose can be placed in the TPN bag as regular insulin [2]
-
Correctional doses can be given subcutaneously for unusual hyperglycaemia
-
Avoid long-acting insulin in PN solutions
-
Meal-time insulin should be adjusted according to the carbohydrate content of meals for adequate glycemic control
-
Monitor glucose every 4–6 hours or use CGM [2]
-
Macronutrient goals:
-
Dextrose: ≥2 g/kg/day
-
Lipids: 0.7–1.5 g/kg/day
-
Amino acids: 1.3–1.5 g/kg/day
11.4 Insulin and Snack Coordination
-
Basal insulin patients may require scheduled snacks: midmorning, midday, evening, bedtime [2]
-
Snacks should balance carbohydrates, protein, and fats (e.g., fruit with almonds) [4]
-
Adjust insulin to meal carbohydrate content [2]
11.5 Procedural Fasting and Hypoglycemia Risk
Patients in hospitals frequently undergo procedures such as endoscopy, X-rays, and ECGs, where prolonged fasting or movement within the hospital may increase the risk of hypoglycemia. The nursing team must be well-equipped with knowledge of hypoglycemia management. There should also be glucose meters and supplies for immediate management of hypoglycemia. The food environment is critical especially for elderly patients (4). To prevent such episodes, appropriate snacking with a mix of carbohydrates, protein, and fat—such as whole fruit with almonds—can help maintain glucose levels over time. Hospital-provided snacks should avoid sugar-rich options like sweetened beverages or fruit juices, emphasizing whole fruits and healthy alternatives.
11.6 Special Considerations
-
Elderly and dysphagic patients: Use diabetes-specific formulas; monitor closely for malnutrition [4]
-
Emphasize individualized plans coordinated with insulin regimens
-
Provide nutrition and diabetes education at discharge to support glycemic control post-discharge [3]
11.7 Monitoring Recommendations
-
For patients who are eating: check glucose before meals and at bedtime
-
For NPO or on continuous feeds: check every 4–6 hours or use CGM [2, 3]
Summary of Recommendations
| Area | Recommendation |
| Nutrition Assessment | Evaluate clinical and nutritional needs, determine macronutrient goals |
| Glycemic Targets | Tailored to patient’s condition and risks |
| Oral Feeding | Consistent carbohydrate intake, scheduled meals/snacks |
| Enteral Nutrition | Basal-bolus regimen, regular insulin preferred |
| Parenteral Nutrition | Use regular insulin in TPN post-IV titration; avoid long-acting insulin |
| Monitoring | Point-of-care testing or CGM every 4–6 hrs |
| Team-Based Care | Include dietitians; empower nurses for dynamic insulin adjustment |
| Discharge Planning | Provide education on nutrition and insulin coordination |
References [75, 76, 77, 78, 20, 79]
12 Monitoring and documentation
Effective preoperative documentation in diabetic patients is vital to reduce perioperative risks and optimize glycemic management. Clinicians must document the diabetes type (Type 1, Type 2, or gestational) and assess glycemic control by recording recent HbA1c, targeting <8%. If this is not achievable, preoperative blood glucose should be maintained between 100–180 mg/dL, with consideration of postponing elective surgeries if control is poor [20, 25, 4]. Medication regimens, including insulin and recent changes, should be reviewed to identify risks of hypo- or hyperglycemia [77, 71]. Comorbidities like ischemic heart disease, renal impairment, autonomic neuropathy, and infection risk must also be documented [75]. Preoperative weight optimization is important in obese patients to reduce airway complications [76]. Anesthesia assessments should include relevant investigations and history of diabetic emergencies (DKA, HHS) to guide perioperative care [77, 71]. Despite recommendations, preoperative HbA1c testing rates remain low (52% in the US, 71% in the UK) [81, 82]. Evidence on diabetes severity’s impact on outcomes is mixed, with some studies showing no correlation and others linking elevated HbA1c to poorer outcomes [78, 79]. Frequent glucose monitoring before, during, and after surgery is critical. For non-diabetic patients, continuous monitoring beyond 72 hours may be unnecessary [131]. Careful insulin dose adjustments and documentation of medication changes support glycemic control. Evaluating infection and healing status is also crucial. Meticulous record-keeping ensures effective management and improved surgical outcomes [24].
| History of diabetes | Yes or No |
| If Yes, T1DM or T2DM | Yes or No |
| Gestational diabetes mellitus | Yes or No |
| Duration of diabetes | |
| Evaluate risk for | Ischemic heart disease Autonomic neuropathy Infection, if any Renal complications Diabetes complications |
| Past medication history Medications and dosage |
|
| History of comorbidities | |
| Height, weight and BMI | |
| Blood pressure measurements at admission | |
| Blood glucose levels at admission | Capillary blood glucose HbA1c |
| Electrolytes and acid-base status | |
| Risk factors of hypoglycaemia | Age ≥ 65 years Body mass index ≤ 27 kg/m2 Total daily dose of insulin ≥ 0.6 units/kg (In T2DM) History of stage ≥3 chronic kidney disease (estimated glomerular filtration rate < 60 mL/min/1.73 m2) Liver failure Cerebrovascular accident Active malignancy Pancreatic disorders Congestive heart failure, or infection History of preadmission hypoglycemia or hypoglycemia occurring during a recent or current hospitalization; or impaired awareness of hypoglycemia. |
Recommendations
Monitor blood glucose levels frequently (6-point BG monitoring i.e. fasting, pre-lunch, 2-hour post-lunch, predinner, 2-hour post-dinner, and 3 am) and adjust medications accordingly to maintain stable glycemic control.
Check blood sugar levels frequently, especially before, during and after surgery. There may not be a need to continue inpatient blood glucose monitoring beyond the first 72 hours for non-diabetic hospitalized patients with extended hospitalizations.
Closely track insulin doses to ensure appropriate glycemic control, making adjustments based on blood glucose levels and dietary intake.
Keep track of any medication changes, especially those related to diabetes management, and how they impact blood glucose levels.
Assess the patient's overall condition, including signs of infection, delayed healing, or other complications that can be exacerbated by hyperglycemia.
Documenting post-operative measures
Accurate postoperative documentation of blood glucose levels and insulin doses is vital for monitoring trends and adjusting treatment. Recording medication changes and the patient’s condition, including infections or complications, ensures a complete treatment history. This thorough documentation supports better clinical decisions and targeted postoperative care [132, 133].
Recommendations
Accurately record all blood glucose readings to track trends and inform treatment decisions.
Precisely document insulin doses administered to monitor and adjust therapy effectively.
Clearly record all medication adjustments to maintain an accurate medication history.
Thoroughly document the patient's overall condition to capture relevant factors influencing blood glucose control.
13 Planning discharge from the hospital
Effective discharge planning for hospitalized diabetic patients is multidimensional, requiring early risk assessment, structured in-hospital education, thorough discharge preparation, strong communication with outpatient teams, and post-discharge follow-up. Implementing these guidelines supports safe transitions and sustained glycaemic control. To establish a standardized approach for the discharge planning and post-discharge care of hospitalized patients with diabetes, ensuring glycaemic stability, preventing complications, and promoting patient self-management.
A well-structured discharge plan ensures that patients leave the hospital with the knowledge, tools, and follow-up needed to maintain diabetes control and avoid readmission.
13.1 Instructions and Documentation
Deliver clear, concise discharge instructions in both verbal and written formats to ensure understanding and adherence.
13.2 Follow-Up Appointments
Facilitate timely follow-up with relevant healthcare providers to ensure continuity of care and support during the transition to home.
13.3 Medication and Supplies
Verify that the patient has sufficient medications and supplies for glucose monitoring and insulin administration upon discharge.
13.4 Provider Communication
Ensure that discharge summaries are promptly communicated to the outpatient care team to maintain treatment continuity and coordination.
14 Continuity of Care Strategies
Structured transitional care and strong communication across care settings are key to reducing complications and readmissions in diabetic patients.
14.1 Transitional Care Interventions
Implement support systems such as phone calls, telehealth visits, or home health services to reinforce the discharge plan and provide ongoing care.
14.2 Team-Based Communication
Promote collaboration among multidisciplinary team members to provide consistent and coordinated care across the continuum.
14.3 Patient and Caregiver Engagement
Actively engage patients and their caregivers in the care planning process to foster shared decision-making and improve outcomes.
14.4 Community Resource Linkage
Connect patients to external resources and support systems to address long-term self-care needs and improve quality of life.
14.5 Barrier Mitigation
Identify and address logistical or socioeconomic barriers that may hinder a patient’s ability to manage diabetes effectively at home.
15 Evaluation and Quality Improvement
Continuous evaluation of discharge processes and outcomes enables health systems to identify gaps, measure impact, and drive improvement in diabetic care.
Monitoring Outcomes
Track key indicators such as readmissions and glycaemic control to evaluate the effectiveness of discharge planning strategies.
Patient Feedback
Collect feedback from patients and caregivers to understand their experiences and inform improvements in discharge education and support.
Staff Education
Provide ongoing education and training for healthcare professionals to maintain competency in best practices for diabetes discharge planning and transitional care.
16 Hypoglycaemia management
Hypoglycaemia management Definition: The American Diabetes Association (ADA) defines hypoglycaemia as BG <70 mg/dl. This could present as symptomatic or asymptomatic hypoglycaemia. Severe hypoglycaemia requires assistance of another person to actively administer carbohydrates, glucagon, or take other corrective actions.
| Patient-Related Factors |
| Advanced age |
| Cognitive impairment or altered mental status |
| Poor nutritional intake or malnutrition |
| Inconsistent eating habits, feeding interruptions, or limited access to carbohydrates |
| Long duration of diabetes |
| Disease-Related Factors |
| Chronic kidney disease |
| Congestive heart failure |
| Liver failure or hepatic dysfunction |
| Sepsis or severe infections |
| Active malignancy |
| Treatment-Related Factors |
| Intensive or complex insulin therapy |
| Type 1 diabetes mellitus |
| Use of concomitant medications that increase the risk of hypoglycemia (e.g., sulfonylureas, beta-blockers) |
16.1 Mild Symptoms with BG >70 mg/dL
Patients exhibiting symptoms of hypoglycaemia but with BG >70 mg/dL typically do not require emergency intervention but benefit from small carbohydrate intake to stabilize glucose levels.
| Action: Administer a small carbohydrate snack. |
| Examples: |
| One medium banana |
| A slice of bread or toast |
| A regular meal, if scheduled |
16.2 Confirmed Hypoglycaemia (BG <70 mg/dL)
All patients with BG <70 mg/dL, regardless of symptoms, require immediate treatment to prevent progression.
-
Conscious and Able to Swallow
| Step 1: Treat with 15–20 g of fast-acting carbohydrates: |
| ½ cup (120–150 mL) juice or non-diet soda |
| 3–4 glucose tablets |
| 1 tbsp sugar or honey |
| 6–7 glucose candies |
| Step 2: Recheck BG after 15 minutes |
| If BG remains <70 mg/dL, repeat treatment. |
| If BG ≥70 mg/dL and the next meal is >1 hour away, follow up with a long-acting carbohydrate: |
| Two biscuits |
| A slice of bread/toast |
| A 200–300 mL glass of milk |
16.2.2 Unconscious or Unable to Swallow
-
Do not give anything orally.
| Treatment options: |
| With IV access: 25–50 mL of 50% dextrose (D50) IV bolus |
| Follow with 10% dextrose infusion (D10W) if needed |
| Without IV access: 1 mg glucagon IM or SC |
| Monitor BG every 15 minutes and reassess consciousness |
| Once BG >70 mg/dL and patient is alert, give a long-acting carbohydrate as above |
16.2.3 Special Considerations Based on Insulin Use
ICU Patients on Insulin Infusion: After achieving target BG, resume insulin infusion at half the previous rate.
Patients on Subcutaneous Insulin and NPO: Resume subcutaneous insulin once BG stabilizes, with adjusted dosing if needed.
16.2.4 Ongoing Monitoring and Insulin Therapy
Do not omit insulin doses unless clinically contraindicated, but dose revision may be necessary.
-
Resume insulin once patient stabilizes
-
Continue capillary BG monitoring every 2–4 hours for 24–48 hours
-
Evaluate insulin regimen to prevent recurrence
16.2.5 Documentation and Education
Accurate record-keeping and patient education are key to safe and effective care.
| Document in-patient records | Educate the patient and caregivers | Identify and Address the Cause |
| Time of episode and treatment given Response to treatment Suspected cause and follow-up plan |
Symptoms and treatment of hypoglycaemia When and how to seek help Proper insulin use and glucose monitoring o Carrying fast-acting carbs |
Meal patterns and timing Insulin or medication dosing errors Increased physical activity Alcohol intake Intercurrent illness Renal or hepatic impairment |
| Summary |
| The treatment of hypoglycemia is guided by the ADA definition of hypoglycemia with a cut off plasma glucose concentration of 70 mg/dl. |
| If the patient is conscious, oriented and able to swallow, initial15-20 gm quick acting carbohydrate would suffice, with the monitoring and the step-up approach depending on the response for the increase in the plasma glucose. |
| If the patient is unconscious, IV access or an infusion pump is preferred to provide 100-200 ml of 10% glucose over 15 minutes (e.g. 400-500 ml/hr), with monitoring to repeat till the plasma glucose is 70mg/dl. |
Concluding remarks
To bridge the gap- lending scope for future studies
REFERENCES
1Umpierrez, G. E., Isaacs, S. D., Bazargan, N., You, X., Thaler, L. M., Kitabchi, A. E., “Hyperglycemia: An Independent Marker of In-Hospital Mortality in Patients with Undiagnosed Diabetes,” The Journal of Clinical Endocrinology & Metabolism, vol. 87, p. 978–982, March 2002.
2Dhatariya, K., Mustafa, O. G., Rayman, G., “Safe care for people with diabetes in hospital,” Clinical Medicine, vol. 20, p. 21–27, January 2020.
3Ketan Dhatariya, G. E. U., Management of Diabetes and Hyperglycemia in Hospitalized Patients — ncbi.nlm.nih.gov, 2025.
4Dougherty, S. M., Schommer, J., Salinas, J. L., Zilles, B., Belding-Schmitt, M., Rogers, W. K., Shibli-Rahhal, A., O’Neill, B. T., “Immediate preoperative hyperglycemia correlates with complications in non-cardiac surgical cases,” Journal of Clinical Anesthesia, vol. 74, p. 110375, November 2021.
5Umpierrez, G. E., Hellman, R., Korytkowski, M. T., Kosiborod, M., Maynard, G. A., Montori, V. M., Seley, J. J., Van den Berghe, G., “Management of Hyperglycemia in Hospitalized Patients in Non-Critical Care Setting: An Endocrine Society Clinical Practice Guideline,” The Journal of Clinical Endocrinology & Metabolism, vol. 97, p. 16–38, January 2012.
6Carpenter, D. L., Gregg, S. R., Xu, K., Buchman, T. G., Coopersmith, C. M., “Prevalence and Impact of Unknown Diabetes in the ICU,” Critical Care Medicine, vol. 43, p. e541–e550, December 2015.
7Pasquel, F. J., Lansang, M. C., Dhatariya, K., Umpierrez, G. E., “Management of diabetes and hyperglycaemia in the hospital,” The Lancet Diabetes & Endocrinology, vol. 9, p. 174–188, March 2021.
8Schmeltz, L. R., DeSantis, A. J., Thiyagarajan, V., Schmidt, K., O’Shea-Mahler, E., Johnson, D., Henske, J., McCarthy, P. M., Gleason, T. G., McGee, E. C., Molitch, M. E., “Reduction of Surgical Mortality and Morbidity in Diabetic Patients Undergoing Cardiac Surgery With a Combined Intravenous and Subcutaneous Insulin Glucose Management Strategy,” Diabetes Care, vol. 30, p. 823–828, April 2007.
9Swanson, C. M., Potter, D. J., Kongable, G. L., Cook, C. B., “Update on Inpatient Glycemic Control in Hospitals in the United States,” Endocrine Practice, vol. 17, p. 853–861, November 2011.
10Kar, P., Plummer, M. P., Abdelhamid, Y. Ali, Giersch, E. J., Summers, M. J., Weinel, L. M., Finnis, M. E., Phillips, L. K., Jones, K. L., Horowitz, M., Deane, A. M., “Incident Diabetes in Survivors of Critical Illness and Mechanisms Underlying Persistent Glucose Intolerance: A Prospective Cohort Study,” Critical Care Medicine, vol. 47, p. e103–e111, February 2019.
11Van Ackerbroeck, S., Schepens, T., Janssens, K., Jorens, P. G., Verbrugghe, W., Collet, S., Van Hoof, V., Van Gaal, L. Van, De Block, C., “Incidence and predisposing factors for the development of disturbed glucose metabolism and DIabetes mellitus AFter Intensive Care admission: the DIAFIC study,” Critical Care, vol. 19, 2015.
12Cook, C. B., Kongable, G. L., Potter, D. J., Abad, V. J., Leija, D. E., Anderson, M., “Inpatient glucose control: a glycemic survey of 126 U.S,” hospitals, Journal of Hospital Medicine, vol. 4, November 2009.
13Hulkower, R. D., Pollack, R. M., Zonszein, J., “Understanding hypoglycemia in hospitalized patients,” Diabetes Management, vol. 4, p. 165–176, March 2014.
14Investigators, N. I. C. E.-S. S., “Intensive versus conventional glucose control in critically ill patients,” New England Journal of Medicine, vol. 360, p. 1283–1297, March 2009.
15McDonnell, M. E., Umpierrez, G. E., “Insulin Therapy for the Management of Hyperglycemia in Hospitalized Patients,” Endocrinology and Metabolism Clinics of North America, vol. 41, p. 175–201, March 2012.
16Qaseem, A., Humphrey, L. L., Chou, R., Snow, V., Shekelle, P., “Use of Intensive Insulin Therapy for the Management of Glycemic Control in Hospitalized Patients: A Clinical Practice Guideline From the American College of Physicians,” Annals of Internal Medicine, vol. 154, p. 260–267, February 2011.
17Investigators, N. I. C. E. -S. S., “Hypoglycemia and Risk of Death in Critically Ill Patients,” New England Journal of Medicine, vol. 367, p. 1108–1118, September 2012.
18Levy, N., Hall, G. M., “National guidance contributes to the high incidence of inpatient hypoglycaemia,” Diabetic Medicine, vol. 36, p. 120–121, December 2018.
19Yamada, T., Shojima, N., Noma, H., Yamauchi, T., Kadowaki, T., “Glycemic control, mortality, and hypoglycemia in critically ill patients: a systematic review and network meta-analysis of randomized controlled trials,” Intensive Care Medicine, vol. 43, p. 1–15, September 2016.
20ElSayed, N. A., Aleppo, G., Aroda, V. R., Bannuru, R. R., Brown, F. M., Bruemmer, D., Collins, B. S., Hilliard, M. E., Isaacs, D., Johnson, E. L., Kahan, S., Khunti, K., Leon, J., Lyons, S. K., Perry, M. L., Prahalad, P., Pratley, R. E., Seley, J. J., Stanton, R. C., Gabbay, R. A., 16. Diabetes Care in the Hospital: Standards of Care in Diabetes—2023, Diabetes Care. vol. 46, p. S267–S278, December 2022.
21Y. Handelsman, Z. T. Bloomgarden, G. Grunberger, G. Umpierrez, R. S. Zimmerman, T. S. Bailey, L. Blonde, G. A. Bray, A. J. Cohen, S. Dagogo-Jack, J. A. Davidson, D. Einhorn, O. P. Ganda, A. J. Garber, W. T. Garvey, R. R. Henry, I. B. Hirsch, E. S. Horton, D. L. Hurley, P. S. Jellinger, L. Jovanovič, H. E. Lebovitz, D. LeRoith, P. Levy, J. B. McGill, J. I. Mechanick, J. H. Mestman, E. S. Moghissi, E. A. Orzeck, R. Pessah-Pollack, P. D. Rosenblit, A. I. Vinik, K. Wyne and F. Zangeneh, “American Association of Clinical Endocrinologists and American College of Endocrinology – Clinical Practice Guidelines for Developing A Diabetes Mellitus Comprehensive Care Plan – 2015 — Executive Summary,” Endocrine Practice, vol. 21, p. 413–437, April 2015.
22Jacobi, J., Bircher, N., Krinsley, J., Agus, M., Braithwaite, S. S., Deutschman, C., Freire, A. X., Geehan, D., Kohl, B., Nasraway, S. A., Rigby, M., Sands, K., Schallom, L., Taylor, B., Umpierrez, G., Mazuski, J., Schunemann, H., “Guidelines for the use of an insulin infusion for the management of hyperglycemia in critically ill patients,” Critical Care Medicine, vol. 40, p. 3251–3276, December 2012.
23Moghissi, E. S., Korytkowski, M. T., DiNardo, M., Einhorn, D., Hellman, R., Hirsch, I. B., Inzucchi, S. E., Ismail-Beigi, F., Kirkman, M. S., Umpierrez, G. E., “American Association of Clinical Endocrinologists and American Diabetes Association Consensus Statement on Inpatient Glycemic Control,” Diabetes Care, vol. 32, p. 1119–1131, June 2009
24Davies, M. J., Aroda, V. R., Collins, B. S., Gabbay, R. A., Green, J., Maruthur, N. M., Rosas, S. E., Del Prato, S., Mathieu, C., Mingrone, G., Rossing, P., Tankova, T., Tsapas, A., Buse, J. B., “Management of Hyperglycemia in Type 2 Diabetes, A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD),” Diabetes Care, vol. 45, p. 2753–2786, September 2022.
25Korytkowski, M. T., Muniyappa, R., Antinori-Lent, K., Donihi, A. C., Drincic, A. T., Hirsch, I. B., Luger, A., McDonnell, M. E., Murad, M. H., Nielsen, C., Pegg, C., Rushakoff, R. J., Santesso, N., Umpierrez, G. E., “Management of Hyperglycemia in Hospitalized Adult Patients in Non-Critical Care Settings: An Endocrine Society Clinical Practice Guideline,” The Journal of Clinical Endocrinology & Metabolism, vol. 107, p. 2101–2128, June 2022.
26Malcolm, J., Halperin, I., Miller, D. B., Moore, S., Nerenberg, K. A., Woo, V., Yu, C. H., “In-Hospital Management of Diabetes,” Canadian Journal of Diabetes, vol. 42, p. S115–S123, April 2018.
27Razavi Nematollahi, L. and Omoregie, C., “Updates on the Management of Hyperglycemia in Hospitalized Adult Patients,” Endocrines, vol. 4, p. 521–535, July 2023
28D. J. Magliano, E. J. Boyko and I. D. F. D. A. 10th edition scientific committee, “IDF DIABETES ATLAS,”, 2021.
29Anjana, R. M., Deepa, M., Pradeepa, R., Mahanta, J., Narain, K., Das, H. K., Adhikari, P., Rao, P. V., Saboo, B., Kumar, A., Bhansali, A., John, M., Luaia, R., Reang, T., Ningombam, S., Jampa, L., Budnah, R. O., Elangovan, N., Subashini, R., Venkatesan, U., Unnikrishnan, R., Das, A. K., Madhu, S. V., Ali, M. K., Pandey, A., Dhaliwal, R. S., Kaur, T., Swaminathan, S., Mohan, V., Anjana, R. M., Deepa, M., Pradeepa, R., Mahanta, J., Narain, K., Das, H. K., Adhikari, P., Rao, P. V., Saboo, B., Kumar, A., Bhansali, A., John, M., Luaia, R., Reang, T., Ningombam, S., Jampa, L., Budnah, R. O., Elangovan, N., Subashini, R., Venkatesan, U., Unnikrishnan, R., Das, A. K., Madhu, S. V., Ali, M. K., Pandey, A., Dhaliwal, R. S., Kaur, T., Swaminathan, S., Mohan, V., Sudha, V., Parvathi, S. J., Jayashri, R., Velmurugan, K., Borah, P. K., Rao, S. B., Padhiyar, J. M., Sharma, S., Lalramenga, P., Das, S. K., Singh, T. B., Kaki, T., Basaiawmoit, M. R., Shukla, D. K., Rao, M. N., Joshi, P. P., Dhandania, V. K., Joshi, S. R., Yajnik, C. S., “Prevalence of diabetes and prediabetes in 15 states of India: results from the ICMR-INDIAB population-based cross-sectional study,” The Lancet Diabetes & Endocrinology, vol. 5, p. 585–596, August 2017.
30Mathur, P., Kulothungan, V., Leburu, S., Krishnan, A., Chaturvedi, H. K., Salve, H. R., Amarchand, R., Nongkynrih, B., Kumar, P. G., V.U.K., S., Ramakrishnan, L., Laxmaiah, A., Boruah, M., Kumar, S., Patro, B. K., Raghav, P. R., Rajkumar, P., Sarma, P. S., Sharma, R., Tambe, M., Thankappan, K. R., Arlappa, N., Mahanta, T. G., Joshi, R. P., Rustagi, N., Gupta, S., Behera, B. K., Shelke, S. C., Galhotra, A., Bhuyan, P. J., Pakhare, A. P., Kumar, D., Topno, R. K., Gupta, M. K., Trivedi, A. V., Garg, S., “National noncommunicable disease monitoring survey (NNMS) in India: Estimating risk factor prevalence in adult population,” PLOS ONE, vol. 16, p. e0246712, March 2021.
31Maiti, S., Akhtar, S., Upadhyay, A. K., Mohanty, S. K., “Socioeconomic inequality in awareness, treatment and control of diabetes among adults in India: Evidence from National Family Health Survey of India (NFHS), 2019-2021,” Scientific Reports, vol. 13, February 2023.
32Ranasinghe, P., Jayawardena, R., Gamage, N., Sivanandam, N., Misra, A., “Prevalence and trends of the diabetes epidemic in urban and rural India: A pooled systematic review and meta-analysis of 1.7 million adults,” Annals of Epidemiology, vol. 58, p. 128–148, June 2021.
33ADA, “15. Diabetes Care in the Hospital: Standards of Medical Care in Diabetes—2020,” Diabetes Care, vol. 43, p. S193–S202, December 2019.
34Society, J. B. D., Joint British Diabetes Societies (JBDS) for Inpatient Care Group | The Association of British Clinical Diabetologists — abcd.care, 2025.
35Panikar, V., Sosale, A., Agarwal, S., Unnikrishnan, A., Kalra, S., Bhattacharya, A., Chawla, M., Anjana, R. M., Bhatt, A., Jaggi, S., Sosale, B., Hasnani, D., Vadgama, J., “RSSDI clinical practice recommendations for management of In-hospital hyperglycaemia—2016,” International Journal of Diabetes in Developing Countries, vol. 36, p. 1–21, November 2016.
36U.G.E. Dhatariya, K, Management of Diabetes and Hyperglycemia in Hospitalized Patients — ncbi.nlm.nih.gov, 2025.
37Silva-Perez, L. J., Benitez-Lopez, M. A., Varon, J., Surani, S., “Management of critically ill patients with diabetes,” World journal of diabetes, vol. 8, no. 3, p. 89–96, March 2017.
38ElSayed, N. A., Aleppo, G., Bannuru, R. R., Bruemmer, D., Collins, B. S., Ekhlaspour, L., Galindo, R. J., Hilliard, M. E., Johnson, E. L., Khunti, K., Lingvay, I., Matfin, G., McCoy, R. G., Perry, M. L., Pilla, S. J., Polsky, S., Prahalad, P., Pratley, R. E., Segal, A. R., Seley, J. J., Stanton, R. C., Gabbay, R. A., “16. Diabetes Care in the Hospital: Standards of Care in Diabetes—2024, ” Diabetes Care. vol. 47, p. S295–S306, December 2023.
39Alshaya, A., Szumita, P., DeGrado, J., Lupi, K., “Research Snapshot Theater: Endocrine I, ” in Critical Care Medicine, 2020.
40Magaji, V., Johnston, J. M., “Inpatient Management of Hyperglycemia and Diabetes,” Clinical Diabetes, vol. 29, p. 3–9, January 2011.
41Avanzini, F., Marelli, G., Donzelli, W., Busi, G., Carbone, S., Bellato, L., Colombo, E. L., Foschi, R., Riva, E., Roncaglioni, M. C., De Martini, M., “Transition From Intravenous to Subcutaneous Insulin,” Diabetes Care, vol. 34, p. 1445–1450, June 2011.
42Shomali, M. E., Herr, D. L., Hill, P. C., Pehlivanova, M., Sharretts, J. M., Magee, M. F., “Conversion from Intravenous Insulin to Subcutaneous Insulin After Cardiovascular Surgery: Transition to Target Study,” Diabetes Technology & Therapeutics, vol. 13, p. 121–126, February 2011.
43Kreider, K. E., Lien, L. F., “Transitioning Safely from Intravenous to Subcutaneous Insulin,” Current Diabetes Reports, vol. 15, March 2015.
44Thammakosol, K., Sriphrapradang, C., “Effectiveness and safety of early insulin glargine administration in combination with continuous intravenous insulin infusion in the management of diabetic ketoacidosis: A randomized controlled trial,” Diabetes Obesity and Metabolism, vol. 25, p. 815–822, December 2022.
45Hsia, E., Seggelke, S., Gibbs, J., Hawkins, R. M., Cohlmia, E., Rasouli, N., Wang, C., Kam, I., Draznin, B., “Subcutaneous Administration of Glargine to Diabetic Patients Receiving Insulin Infusion Prevents Rebound Hyperglycemia,” The Journal of Clinical Endocrinology & Metabolism, vol. 97, p. 3132–3137, September 2012.
46Lim, Y., Ohn, J. H., Jeong, J., Ryu, J., Kim, S.-w, Cho, J. H., Park, H.-S., Kim, H. W., Lee, J., Kim, E. S., Kim, N.-H., Jo, Y. H., Jang, H. C., “Effect of the Concomitant Use of Subcutaneous Basal Insulin and Intravenous Insulin Infusion in the Treatment of Severe Hyperglycemic Patients,” Endocrinology and Metabolism, vol. 37, p. 444–454, June 2022.
47Zhou, K., Buehler, L. A., Zaw, T., Bena, J., Lansang, M. C., “Weight-Based Insulin During and After Intravenous Insulin Infusion Reduces Rates of Rebound Hyperglycemia When Transitioning to Subcutaneous Insulin in the Medical Intensive Care Unit,” Endocrine Practice, vol. 28, p. 173–178, February 2022.
48Barker, H. L., Morrison, D., Llano, A., Sainsbury, C. A. R., Jones, G. C., “Practical Guide to Glucocorticoid Induced Hyperglycaemia and Diabetes,” Diabetes Therapy, vol. 14, p. 937–945, March 2023.
49Liu, X.-x, Zhu, X.-m, Miao, Q., Ye, H.-y, Zhang, Z.-y, Li, Y.-m, “Hyperglycemia Induced by Glucocorticoids in Nondiabetic Patients: A Meta-Analysis,” Annals of Nutrition and Metabolism, vol. 65, p. 324–332, 2014.
50Bus, S. A., Sacco, I. C. N., Monteiro-Soares, M., Raspovic, A., Paton, J., Rasmussen, A., Lavery, L. A., van Netten, J. J., “Guidelines on the prevention of foot ulcers in persons with diabetes (IWGDF, 2023 update),” Diabetes/Metabolism Research and Reviews, vol. 40, June 2023.
51Bell, S., Ross, V. C., Zealley, K. A., Millar, F., Isles, C., “Management of post-operative acute kidney injury,” QJM, hcw175, October 2016.
52Arun, B. G., Chandrashekar, S., Goyal, K., ““Power” ful eye causing postoperative blurring of vision after non-ocular surgery under general anesthesia,” Journal of Anaesthesiology Clinical Pharmacology, vol. 39, p. 141–142, June 2022.
53Kitabchi, A. E., Umpierrez, G. E., Miles, J. M., Fisher, J. N., “Hyperglycemic Crises in Adult Patients With Diabetes,” Diabetes Care, vol. 32, p. 1335–1343, July 2009.
54Lizzo, J. M., Goyal, A., Gupta, V., “Adult Diabetic Ketoacidosis — ncbi.nlm.nih.gov, 2023.
55K. A. E. Gosmanov, A. R., Diabetic Ketoacidosis — ncbi.nlm.nih.gov, 2018.
56Taylor, S. I., Blau, J. E., Rother, K. I., “SGLT2 Inhibitors May Predispose to Ketoacidosis,” The Journal of Clinical Endocrinology & Metabolism, vol. 100, p. 2849–2852, August 2015.
57Jung, Y., Lau, A., Bednarczyk, J., “Immune checkpoint inhibitor-associated diabetic ketoacidosis and insulin-dependent diabetes: a case report,” Journal of Medical Case Reports, vol. 18, December 2024.
58Kitabchi, A. E., Umpierrez, G. E., Murphy, M. B., Barrett, E. J., Kreisberg, R. A., Malone, J. I., Wall, B. M., “Management of Hyperglycemic Crises in Patients With Diabetes,” Diabetes Care, vol. 24, p. 131–153, January 2001.
59Fayfman, M., Pasquel, F. J., Umpierrez, G. E., “Management of Hyperglycemic Crises,” Medical Clinics of North America, vol. 101, p. 587–606, May 2017.
60Dhatariya, K. K., “The management of diabetic ketoacidosis in adults—An updated guideline from the Joint British Diabetes Society for Inpatient Care,” Diabetic Medicine, vol. 39, February 2022.
61Gerich, J. E., “Role of the kidney in normal glucose homeostasis and in the hyperglycaemia of diabetes mellitus: therapeutic implications,” Diabetic Medicine, vol. 27, p. 136–142, February 2010.
62Pecoits-Filho, R., Abensur, H., Betônico, C. C. R., Machado, A. D., Parente, E. B., Queiroz, M., Salles, J. E. N., Titan, S., Vencio, S., “Interactions between kidney disease and diabetes: dangerous liaisons,” Diabetology & Metabolic Syndrome, vol. 8, July 2016.
63Hahr, A. J., Molitch, M. E., “Management of diabetes mellitus in patients with chronic kidney disease,” Clinical Diabetes and Endocrinology, vol. 1, June 2015.
64Chun, J., Strong, J., Urquhart, S., “Insulin Initiation and Titration in Patients With Type 2 Diabetes,” Diabetes Spectrum, vol. 32, p. 104–111, May 2019.
65Freedman, B. I., “A Critical Evaluation of Glycated Protein Parameters in Advanced Nephropathy: A Matter of Life or Death,” Diabetes Care, vol. 35, p. 1621–1624, June 2012.
66Coelho, S., Rodrigues, A., “Hemoglobin A1c in Patients on Peritoneal Dialysis: How Should We Interpret It?,” Therapeutic Apheresis and Dialysis, vol. 18, p. 375–382, February 2014.
67Hoshino, J., Abe, M., Hamano, T., Hasegawa, T., Wada, A., Ubara, Y., Takaichi, K., Nakai, S., Masakane, I., Nitta, K., “Glycated albumin and hemoglobin A1c levels and cause-specific mortality by patients’ conditions among hemodialysis patients with diabetes: a 3-year nationwide cohort study,” BMJ Open Diabetes Research & Care, vol. 8, p. e001642, October 2020.
68Tsuruta, Y., Ichikawa, A., Kikuchi, K., Echida, Y., Onuki, T., Nitta, K., “Glycated albumin is a better indicator of the glucose excursion than predialysis glucose and hemoglobin A1c in hemodialysis patients,” Renal Replacement Therapy, vol. 2, January 2016.
69ADA, “15. Diabetes Care in the Hospital: Standards of Medical Care in Diabetes—2021,” Diabetes Care, vol. 44, p. S211–S220, December 2020.
70Rajendran, R., Kerry, C., Rayman, G., “Temporal patterns of hypoglycaemia and burden of sulfonylurea-related hypoglycaemia in UK hospitals: a retrospective multicentre audit of hospitalised patients with diabetes,” BMJ Open, vol. 4, p. e005165, July 2014.
71Roberts, A., James, J., Dhatariya, K., “Management of hyperglycaemia and steroid (glucocorticoid) therapy: a guideline from the Joint British Diabetes Societies (JBDS) for Inpatient Care group,” Diabetic Medicine, vol. 35, p. 1011–1017, July 2018.
72>U. G. E. Dhatariya K, Management of Diabetes and Hyperglycemia in Hospitalized Patients. [Updated 2024 Oct 20]. In: Feingold KR, Ahmed SF, Anawalt B, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279093/, A. B. e. a. e. E. [. S. D. (. M. I. n: Feingold KR and 2000-., Eds., MDText.com, Inc.; 2000, 2024.
73A.S.P.E.N. Clinical Guidelines: Nutrition Support of Adult Patients With Hyperglycemia,” Journal of Parenteral and Enteral Nutrition, vol. 37, p. 23–36, June 2012.
74Lobo, D. N., Gianotti, L., Adiamah, A., Barazzoni, R., Deutz, N. E. P., Dhatariya, K., Greenhaff, P. L., Hiesmayr, M., Jakobsen, D. Hjort, Klek, S., Krznaric, Z., Ljungqvist, O., McMillan, D. C., Rollins, K. E., Sekeljic, M. Panisic, Skipworth, R. J. E., Stanga, Z., Stockley, A., Stockley, R., Weimann, A., “Perioperative nutrition: Recommendations from the ESPEN expert group,” Clinical Nutrition, vol. 39, p. 3211–3227, November 2020.
75Gosmanov, A. R., Umpierrez, G. E., “Medical Nutrition Therapy in Hospitalized Patients with Diabetes,” Current Diabetes Reports, vol. 12, p. 93–100, October 2011.
76Whitham, D., “Nutrition management of diabetes in acute care,” Canadian journal of diabetes, vol. 38, no. 2, p. 90–93, April 2014.
77Patzek, S., MacMaster, H. W., Rov-Ikpah, E., Luis, C. San, Johnson, C., Juttukonda, V., Rushakoff, R. J., “Automated Self-Adjusting Subcutaneous Insulin Algorithm for Patients NPO or on TPN or Enteral Feedings,” Journal of Diabetes Science and Technology, vol. 15, p. 1282–1289, August 2020.
78Roberts, H. C., “Changing the food environment: the effect of trained volunteers on mealtime care for older people in hospital,” Proceedings of the Nutrition Society, vol. 77, p. 95–99, October 2017.
79Powers, M. A., Bardsley, J. K., Cypress, M., Funnell, M. M., Harms, D., Hess-Fischl, A., Hooks, B., Isaacs, D., Mandel, E. D., Maryniuk, M. D., Norton, A., Rinker, J., Siminerio, L. M., Uelmen, S., “Diabetes Self-management Education and Support in Adults With Type 2 Diabetes: A Consensus Report of the American Diabetes Association, the Association of Diabetes Care & Education Specialists, the Academy of Nutrition and Dietetics, the American Academy of Family Physicians, the American Academy of PAs, the American Association of Nurse Practitioners, and the American Pharmacists Association,” Diabetes Care, vol. 43, p. 1636–1649, June 2020 .
80Lazar, H. L., McDonnell, M., Chipkin, S. R., Furnary, A. P., Engelman, R. M., Sadhu, A. R., Bridges, C. R., Haan, C. K., Svedjeholm, R., Taegtmeyer, H., Shemin, R. J., “The Society of Thoracic Surgeons Practice Guideline Series: Blood Glucose Management During Adult Cardiac Surgery,” The Annals of Thoracic Surgery, vol. 87, p. 663–669, February 2009 .
81Jason, C. J., Samuhel, M. E., Glick, B. J., Welsh, A. K., “Geographic distribution of unexplained low birth weight,” Journal of occupational medicine. : official publication of the Industrial Medical Association, vol. 28, no. 8, p. 728–740, August 1986.